APPLICATIONS
Lipid Nanoparticles Formulation
Offering multiple technology solutions for Lipid Nanoparticle (LNP) manufacturing processing
LNPs, including various lipid-based platforms such as liposomes, nanostructured lipid carriers (NLCs), and solid lipid nanoparticles (SLNs), are critical for the delivery of protein and amino acid-type antigens.
From R&D all the way up to production scale it is critical to use the best, most suitable methodology. Microfluidics is delighted to be partnering with Inside Therapeutics to extend our range of equipment solutions and help our customers to reach their LNP goals.
YOUR CHALLENGES
Achieving Homogeneous Lipid Nanoparticle Formulations
The challenge in LNP production is manufacturing lipid-based nanoparticles which exhibit strong efficacy and functionality, that not only can be repeated batch-to-batch but also be scaled from lab and/or pilot size to mass-production volumes.
Choosing the right methodology for LNP manufacturing is critical. Are you clear on which is most suitable method for your LNP formulation - a top-down or bottom-up approach? We are here to help you.
HOW WE CAN HELP
Technology for Manufacturing Lipid Nanoparticles
Microfluidics is delighted to add the TAMARA nanoparticle formulation system to the portfolio. An innovative platform that covers two R&D stages from screening to in-vivo scale in one machine, using microfluidic formulation equipment.
We are the only company to offer both top-down & bottom-up LNP mixing equipment. Our equipment is simple to use, gives reliable, superior results and the ability to scale using Microfluidizer® technology.
OUR APPROACH
Reproducible Lipid Nanoparticle Manufacturing
Microfluidizer® technology is a trusted top-down solution for lipid nanoparticle production. The equipment is easy to operate and produces consistent particle sizes with narrow distributions.
The TAMARA platform adds R&D scale, bottom-up technology to the portfolio. This simple to use, plug & play module can process samples in under 2 minutes. Reusable microfluidic chips make it a sustainable, cost-effective choice. Our team will help you find the best equipment for lipid nanoparticle manufacturing.
OUR EXPERIENCE
Scalable Production of Lipid Nanoparticles
Bringing TAMARA into our portfolio means that we can offer customers the ability to compare top-down or bottom-up LNP production equipment.
We offer the ability to scale up to production volumes and help deliver your product to the market with Microfluidizer® technology.
Our dedicated team of application engineers & subject matter experts offer support throughout your journey.
TECHNIQUES
Lipid Nanoparticle Formulation
Bottom-up Approach
The most common nanoparticle formulation method relies on the fast mixing of two miscible phases (lipids/polymers in an organic solvent and aqueous buffer), leading to a drop in solubility of the lipids/polymers, which start to agglomerate to form nanoparticles. Commonly known as nanoprecipitation/self-assembly process.
Mixing speed affects the nanoparticle size - faster mixing produces smaller particles. Also, the more efficient and homogeneous the mixing is, the better the control over the size and distribution.
Microfluidic chip-based platforms can maintain the fluid flow in laminar conditions, and provide optimal control of the final nanoparticle physicochemical parameters.
.webp?width=252&height=155&name=TAMARA-11-2024-inside-therapeutics-800-800x494%20(1).webp)
Benefits:
- Fast processing time
- Low volume range µL to mL
- Size control & repeatability
- PDI <0.2 & EE >95%
Top Down Approach
The phospholipid, carrier oil and any hydrophobic actives are dissolved in a solvent, which is subsequently removed via evaporation. A buffer is added to the resulting precipitate and then warmed and vortexed to hydrate the phospholipids.
The antigen (or other hydrophilic actives) are added to the solution whereupon large multi-layer vesicles (MLVs) are generated.
These MLVs are then processed through a Microfluidizer® processor to reduce their size to small uni-lamellar vesicles.
Benefits:
- Scalability with repeatability
- Higher volume range mL to L
- Good size control
- GMP level equipment
RESOURCE
RNA Vaccine Delivery Systems Using LNPs
For more information, view our application note on creating RNA vaccine delivery systems.
This application note compares the in-situ encapsulation method to the post adsorption approach of LNP manufacturing. It is concluded that the post absorption approach is highly suitable for large-scale manufacturing & responding to pandemic situations.

RESULTS
Proven Solutions for LNPs
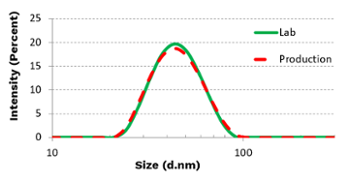
Microfluidizer® technology has been proven as a seamless path for scaling up LNP production. A pharmaceutical company successfully created the ideal liposomes to treat carcinomas.
Small and Uniform Particles
The average particle size was reduced to around 40 nm. The polydispersity index (PdI) was <0.1, indicating high particle size uniformity.
Process Scalability
The process was scaled up directly from lab scale to the production scale. The results obtained from the production scale Microfluidizer® Processor were almost identical to the results produced by the lab machine (see graph).
RESOURCE
Streamline mRNA-LNP Screening With TAMARA
When conducting the initial research steps, it is important to get the screening right. The bench-scale system, TAMARA, provides control over particle size, Polydispersity index (PDI), and Encapsulation efficiency (EE%).
Read the Application Note to explore how to streamline your screening methods.

RESOURCES
Lipid Nanoparticles Information
- Using lipid nanoparticles for mRNA delivery
- A scalable solution for next generation RNA technology and delivery
- Using lipid nanoparticles to design vaccine delivery systems
- FAQ: How does Microfluidizer technology support vaccine development?
- Technology for Cancer Drug Delivering Nanoparticle Manufacturing




