CBD Oil & Hemp Processing Equipment
Microfluidizer® High Shear Homogenizer Technology
for creating next generation products.
High Shear Homogenizers for Processing CBD Oil and Hemp
Now that Cannabis and Hemp-based products are moving into the mainstream market place, it is more important than ever to develop safe, effective, and consistent products.
Creating Next-Generation Cannabis Products
A major challenge for manufacturers is to formulate Hemp oil and CBD oil in such a way as to make them usable in consumable products.This is because of the following:
- The oil is poorly water-soluble, and it takes a lot of high-intensity mixing to achieve shelf-stable preparations.
- In its natural, extracted form, CBD oil has a low bioavailability - typically 6-20%(1) by direct oral administration of raw or unencapsulated cannabis oil.
- Cannabis products that are not carefully manufactured show a lot of variability in effectiveness. The onset of the effect can be delayed as much as 60 minutes, and it takes hours after digestion to reach peak effect. These deficiencies can make the product appear weak and lead to over-consumption or cause increased adverse effects(2).
The issue of low bioavailability has been overcome in the
pharmaceutical industry by producing nanoemulsions or other nanoencapsulations such as liposomes. These nano-formulations have been demonstrated to:
- Increase bioavailability by as much as 50-75%(1)
- Reduce response time.
- Lower the doses needed to achieve similar effects - this lower dosage reduces the potential occurrence of adverse effects.
- Reduce the amount of active ingredient required to achieve the same physiological effect.
- Mask the taste of the CBD oil and Hemp oil.
- Produce droplet sizes less than 100nm which makes the formulation translucent or even transparent in appearance which is a desirable aesthetic quality when trying to create clear cannabis water.
- Increase stability of the formulation and produce a longer shelf-life.
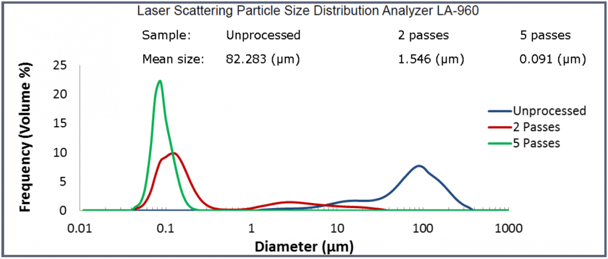
Microfluidizer® Processor High Shear Homogenizers for Processing CBD Oil
The Microfluidizer® processor technology is the only technology that can create nano-sized emulsions or encapsulations (liposomes) that meet all of the above requirements.
The Microfluidizer® processor provides the highest uniform shear rates on the market by utilizing
a fixed geometry Interaction ChamberTM and extremely high constant processing pressures. The
Microfluidizer® technology is also guaranteed to scale up so that product processing and results are consistent from R&D development to manufacturing. In addition, Microfluidizer® processors have a
proven track record for meeting FDA requirements for food and pharmaceutical production and have the capability to be cGMP compliant.
Molecular weight reduction of cannabis oil is common practice. This technique reduces the viscosity and makes the oil easier to work with in delivery methods such as vaporizers. Chemical processing modifies the chemical structure of cannabinoids and the chemicals add cost and undesirable contaminants, which increases the requirements for approval, testing and regulation. A Microfluidizer® processor is superior to a chemical approach because it does not require any chemicals in the process and will not alter the cannabinoids.
For producers, extractors, and processors who want to improve CBD and cannabis-related products, the Microfluidizer® high shear processors provide a scalable solution with the highest efficiency. The resulting unique formulations will differentiate your products with improved shelf life, stability, and effectiveness.
IDEX Material Processing Technologies (IDEX MPT)
Microfluidics™, part IDEX Material Processing Technologies (IDEX MPT), delivers a range of processing technologies for market-leading products. This platform includes: Fitzpatrick Hammer Milling for Hemp Processing, Quadro Engineering Milling Equipment and High Shear Mixing Equipment.
Please note - Microfluidics information and literature is intended only for the use of its equipment in jurisdictions where such activities are legal.
REFERENCES AS MENTIONED IN TEXT ABOVE:
(1) Goodwin R., Gustafson R.A., Barnes A., Nebro W., Moolchan E.T. $ Huestis M.A.(2006) - ∆9-tetrahydrocannabinol, 11-hydroxy-∆9-tetrahydrocannabinol and 11-nor-9-carboxy-∆ 9-tetrahydrocannavinol in human plasma after controlled oral administration of cannabinoids. [Ther Drug Monit. 24(4): pp 545-51]
(2) Lemberger L.,Weiss J.L., Watanabe A.M., Galanter I.M., Wyatt R.J., & Cardon P.V.(1972) - Delta-9 tetrahydrocannabinol: temporal correlation of the psychologic effects and blood levels after various routes of administration [N Engl J of Med. 286(13): pp 685-88]


