Although the world’s attention focuses on developing and distributing viable Covid-19 vaccines in warp speed, we shouldn’t ignore that rapid and accurate diagnostic testing continues to be a priority in stopping the spread. In the early days of the virus, inadequate availability, inaccurate results and long turnaround times were common complaints of COVID-19 testing. Lengthy wait periods between testing and results often took weeks and false negative test results were reported at approximately 20%, subject to the time of testing1. Identifying and testing this virus has been challenging but accurate diagnostic testing, with improved turnaround times, are instrumental in stopping the spread of Covid-19.
Overview of COVID-19 Diagnostic Testing
Most molecular diagnostic testing, such as Covid-19 testing, relies on the process of amplification which entails duplication of RNA and DNA sequences that are used for detection and diagnosis. Through amplification, a multitude of DNA replications are harvested, detected, and measured. In the diagnostic community, Polymerase Chain Reaction (PCR) is considered the gold standard of diagnostic testing due to its ability to create unlimited copies from just one strand of DNA. Now an alternative Covid-19 diagnostic testing method has proven to be both highly sensitive and highly specific and is available based on the Clustered Regularly Interspaced Short Palindromic Repeats (CRISPR) technology.2-4
CRISPR Technology
CRISPR is a gene targeting and gene editing technology which is providing a more specific and targeted approach at detection. The CRISPR method allows for the detection of specific COVID-19 nucleic acid sequences when associated with Cas12 or Cas13 enzymes. A visual signal can then be produced with the help from reporter molecules which is activated upon target recognition.
The CRISPR-Cas detection method, in conjunction with the Loop-mediated Isothermal Amplification (LAMP) method, is found to be a rapid and reliable diagnostic test. Unlike PCR, LAMP does not use a heat cycle and therefore results are faster and require less equipment. Previous limitations of LAMP resulted in false positive results due to its isothermal nature and replication of off target sequences. CRISPR technology now helps to address these problems, resulting in fast and reliable results.
The CRISPR-based testing, has shown:
- Faster turnaround from patient application to test results.
- Fewer instruments needed in the test lab to analyze the sample.
- High accuracy of test results – both positive and negative.
- Potential over-the-counter availability with easy comprehension of results.
So What is Microfluidics Role in CRISPR Diagnostic Testing?
Just like the Taq polymerase in PCR, Cas proteins are essential elements for the CRISPR-based diagnostic method. They target specific sites on viral DNA or RNA and release a signal which is later analyzed to interpret test results. These enzymes are often engineered and expressed in bacterial cells such as E. Coli. Extraction of these enzymes via cell rupture is key in developing diagnostic COVID-19 tests and mass production of test kits.
The Microfluidizer® processors can be used effectively in producing those critical enzymes for both PCR and CRISPR-based diagnostic methods. The challenge in cell disruption is not only lysing cells safely, so the intracellular enzymes are released undamaged, but also maintaining stable and repeatable results batch-to-batch. Microfluidizer® technology generates superior cell rupture results by achieving high yields in enzyme recovery through controlled shear force. Each batch receives the same treatment, resulting in more uniform results and easier downstream filtration which are key elements in cell disruption.
The Microfluidizer® processor meets and often exceeds other high pressure homogenizers (HPH) because:
- It offers fast results and samples are processed in shorter times than alternative methods
- It is easy to clean and requires little maintenance
- Its cooling system is efficient as not to risk damaging sensitive biological cell contents
- It gives an overall better separation of cell protein from debris
- It provides lower viscosity for easier filtration
Due to the incorporation of the revolutionary multi-channeled Interaction Chamber™, the Microfluidizer® technology has scale up capabilities ensuring results are uniform each time regardless of volume. Therefore, results are the same from lab to pilot to production.
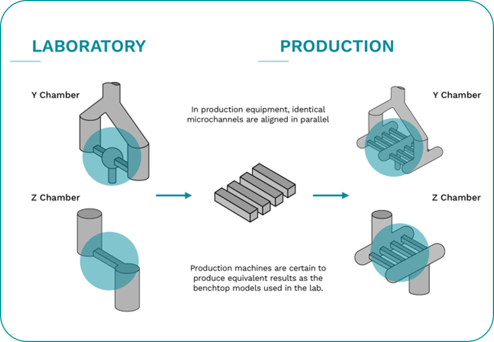
In the fight against COVID-19, Microfluidics offers unparalleled results and offers years of expertise in solving companies’ most complex issues. From research to manufacturing, Microfluidics has superior knowledge to help achieve superior results.
References
Topics:
Cell Disruption
