Application Note
Comparison of Methods for Formulating RNA-LNPs
This study assesses TAMARA, a microfluidic system for formulating RNA-loaded lipid nanoparticles at very low volumes, to reduce RNA and reagent use while maintaining consistent nanoparticle quality and biological performance.
Read the application note to see the results of the study comparing TAMARA with an established toroidal mixer system.

Application
RNA-LNP Formulation
Industry
Biopharma
Key Products
ABSTRACT
What are the common methods for formulating RNA-LNP?
Screening RNA-loaded lipid nanoparticle (RNA-LNP) formulations is essential for developing efficient mRNA vaccines and therapeutics. Microfluidic systems are now the preferred approach, as they minimise reagent use while giving precise control over critical nanoparticle properties.
This study evaluates the TAMARA nanoparticle formulation system, with a particular focus on its performance at low working volumes. The aim is to optimize RNA-LNP formulations while reducing consumption of expensive mRNA. TAMARA is also directly compared with a widely used toroidal mixer to clearly assess the capabilities of both systems.
Both systems generated high-quality RNA-LNPs with optimal size and PDI. However, TAMARA shows strong potential as an alternative to the toroidal mixer, especially in terms of encapsulation efficiency, virtually eliminating reagent losses and achieving higher protein expression levels. By reducing reagent usage, maintaining performance, and enabling chip reusability, TAMARA offers a cost-effective and practical platform for advancing RNA-LNP therapeutic development.
| Key Elements | TAMARA | Toroidal Mixer |
| mRNA-LNP Formulation System Tested | TAMARA system by Inside Therapeutics | Ignite by Precision Nanosystems |
| Formulation Volumes | 200 µL / 400 µL / 700 µL | 700 µL |
| Flow Parameters | TFR, FRR | Previously optimized |
| Physiochemical Characterization |
Particle Size & PDI: Measured using DLS |
Particle Size & PDI: Measured using DLS EE% and EY%: RiboGreen assay |
| In-Vitro Studies | Transcription Efficiency: RNA delivery efficacy Cell Viability: Cytotoxicity Cells Fluorescence Intensity: Protein expression levels |
Transcription Efficiency: RNA delivery efficacy Cell Viability: Cytotoxicity Cells Fluorescence Intensity: Protein expression levels |
INTRODUCTION
A comparison of methods for formulating RNA-LNPs
Formulating RNA-loaded lipid nanoparticles (RNA-LNPs) is central to developing mRNA vaccines and therapeutics, as they protect RNA and enable efficient delivery into cells. However, creating robust mRNA-LNP formulations is challenging. Researchers must screen many lipid compositions and process conditions at low volumes to identify the best candidates for in vivo studies and clinical development.
Microfluidic systems have become a preferred solution for RNA-LNP formulation. They enable low-volume processing that reduces consumption of costly RNA while maintaining tight control over key nanoparticle attributes, such as particle size, PDI, and encapsulation efficiency.
This application note evaluates TAMARA, a microfluidic-based nanoparticle formulation system, at low working volumes of 200 µL, 400 µL, and 700 µL. The objective is to understand how effectively TAMARA can minimize reagent use while maintaining consistent performance across critical RNA-LNP parameters, including particle size, encapsulation efficiency and yield, transcription efficiency, cell viability, and protein expression levels.
In a second phase, TAMARA’s performance is benchmarked against a widely used formulation system with a toroidal mixer. This comparator system, previously optimized using a Design of Experiments (DOE) approach, provides a robust reference for assessing TAMARA.
The results offer insight into the advantages of TAMARA for low-volume RNA-LNP screening, particularly its ability to deliver virtually zero material losses and precise volume control. These advantages support more cost‑effective development of RNA-based therapies.
This is a preliminary study. While the data are promising, further detailed validation and comprehensive characterization will be required to fully substantiate these findings.
This work was carried out by the ART “ARNm” team, an Inserm R&D laboratory led by Prof. Chantal Pichon in Orléans, with major contributions from Dr. Nabila Laroui and Dr. Milab Baroud, and support from Inside Therapeutics.
RESULTS
Core insights
A comprehensive physicochemical characterization and in vitro testing were completed to assess the performance of both systems. In the figures and tables that follow, samples are labeled using the following naming format:
For TAMARA:-
- TAM FRR-TFR Volume (in µL)
- Example: For a formulation with an FRR of 4:1, a TFR of 9 mL/min, and a total formulation volume of 700 µL, the sample is labeled as: TAM 4-9 700.
- IGNITE X 700 (where X corresponds to the sample number previously optimized through a DOE and 700 corresponding the 700 µL formulation).
RNA-LNP size & PDI with TAMARA
A: Impact of formulation parameters
This initial study evaluates how different formulation settings affect the size and polydispersity index (PDI) of RNA-LNPs produced with the TAMARA system. Four conditions were tested at a total formulation volume of 700 µL:
- TFR 9 mL/min – FRR 4:1
- TFR 5 mL/min – FRR 4:1
- TFR 9 mL/min – FRR 3:1
- TFR 5 mL/min – FRR 3:1

Figure 1: mRNA-LNP particle size and PDI achieved using TAMARA at a 700 µL formulation volume
As shown in Figure 1, all TAMARA formulations meet industry quality standards, with PDI values below 0.2 and particle sizes in the optimal 80–150 nm range for efficient intracellular delivery.
The data show that increasing the Total Flow Rate (TFR) consistently reduces nanoparticle size, while changes in the Flow Rate Ratio (FRR) have a smaller effect. This makes TFR a straightforward parameter for tuning final nanoparticle size.
These findings are consistent with nanoprecipitation theory: higher flow rates improve mixing between solvent and aqueous phases, creating more nucleation sites and smaller particles. Faster mixing also limits lipid agglomeration and particle coalescence, while higher TFRs increase shear forces within the channels, further preventing the formation of larger particles.
B: Influence of formulation volume
The second study assessed how formulation volume affects nanoparticle size and quality, with the goal of defining the minimum volume that TAMARA can reliably process while maintaining high product quality. Three formulation volumes were tested: 200 µL, 400 µL, and 700 µL. As outlined in the introduction, it was not possible to apply identical formulation conditions across all volumes, so only the flow conditions between volumes were compared.
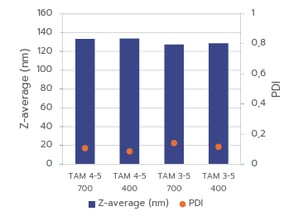
Figure 2(A): Comparison of mRNA-LNP particle size and PDI produced with TAMARA at 400 µL and 700 µL, using the same formulation conditions.
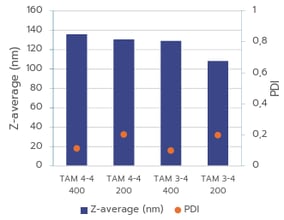
(B): Comparison of mRNA-LNP particle size and PDI generated with TAMARA at 400 µL and 200 µL, using identical formulation conditions.
Figure 2(A) shows excellent consistency in particle size for formulations produced at 700 µL and 400 µL, demonstrating strong scalability between these volumes. However, the PDI increases at lower volumes. Figure 2(B) illustrates that nanoparticle sizes at 400 µL and 200 µL differ significantly, indicating that scaling between these smaller volumes is more complex and will require further optimisation. In addition, formulations at 200 µL show a higher PDI, reflecting a more heterogeneous nanoparticle population. This is expected, as smaller batch volumes are typically more difficult to control and tend to yield broader size distributions and therefore higher PDI values.
Despite these challenges, TAMARA consistently produced high-quality nanoparticles at volumes down to 400 µL. This suggests that 400 µL is a practical lower limit for initial screening, with the opportunity to achieve even better PDI performance at higher formulation volumes.
C: Comparison between TAMARA and optimized toroidal mixer
Finally, based on the findings above, nanoparticles produced with TAMARA at 700 µL and 400 µL were compared with nanoparticles generated with a toroidal mixer operating under pre-optimized conditions.
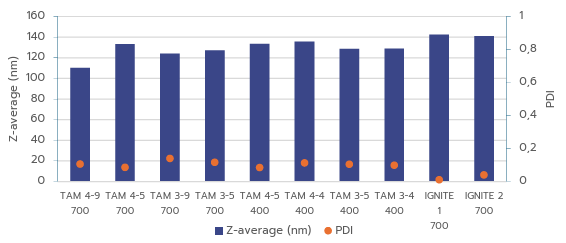
Figure 3: Comparison of mRNA-LNP particle size and PDI for TAMARA across all volumes
and conditions, versus IGNITE under optimized conditions.
Figure 3 shows that RNA-LNPs produced with both systems meet high quality standards, with particle size in the optimal range and PDI below 0.2, in line with regulatory requirements. For the TAMARA system, applying a Design of Experiments (DOE) approach could further reduce PDI, as evidenced by the lower PDI values achieved with the optimized IGNITE formulation.
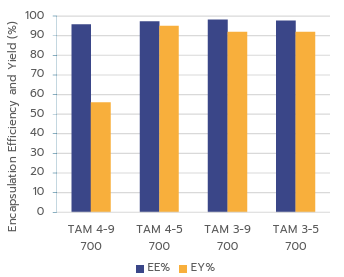
Figure 4: mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) achieved using TAMARA at a 700 µL formulation volume.
 Figure 5(A): Comparison of mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) using TAMARA at 400 µL and 700 µL formulation volumes under the same process conditions.
Figure 5(A): Comparison of mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) using TAMARA at 400 µL and 700 µL formulation volumes under the same process conditions.
 (B): Comparison of mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) using TAMARA at 400 µL and 200 µL formulation volumes under identical process conditions.
(B): Comparison of mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) using TAMARA at 400 µL and 200 µL formulation volumes under identical process conditions.
EE% and EY% study with TAMARA
A: Impact of formulation parameters
After evaluating how formulation parameters affect RNA-LNP size, their impact on encapsulation efficiency (EE%) and encapsulation yield (EY%) were examined. The same formulation conditions used in the size and PDI studies were applied in this analysis.
The data show that formulation parameters have minimal effect on encapsulation efficiency. All RNA-LNPs produced with TAMARA achieved EE% values above 95%, in line with the best-performing systems reported in the literature. TAMARA also delivers exceptional encapsulation yields, up to 95%, which exceeds most previously published results for RNA-LNPs1. The sample labeled TAM 4-9 700 was identified as an outlier.
B: Impact of formulation volume
The impact of total formulation volume on RNA-LNP encapsulation was also assessed using the TAMARA system. As noted above, three volumes (200 µL, 400 µL and 700 µL) were tested under identical formulation conditions.
As shown in Figure 5, encapsulation efficiency (EE%) remains consistently high at all volumes, confirming that TAMARA reliably maximizes RNA-LNP encapsulation. A modest drop in EE% is observed at 200 µL, from 95% at the higher volumes to 85%.
Formulation volume has a greater influence on encapsulation yield (EY%). The lowest volume (200 µL) delivers reduced yield compared with 400 µL and 700 µL. This is likely due to the very small volumes of the two liquid phases (50 µL lipids and 150 µL aqueous at an FRR of 1:3), which produce a very short run time of around 3 seconds. Under these conditions, mixing timing becomes critical, and even slight mismatches can significantly lower yield. In contrast, larger volumes provide longer formulation times, are easier to control and deliver higher yields.
Based on these findings, the Inside Therapeutics team is developing an enhanced version of TAMARA with optimized control of formulation parameters at very low formulation volumes.
C: Comparison between TAMARA & Toroidal mixer
To complete the encapsulation characterization study, the toroidal mixer and the TAMARA system were compared across a range of volumes and formulation parameters.
 Figure 6: Comparison of mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) achieved with TAMARA
Figure 6: Comparison of mRNA-LNP encapsulation efficiency (EE%) and encapsulation yield (EY%) achieved with TAMARA
across all tested volumes and formulation conditions, versus Ignite under its optimized conditions.
Both microfluidic methods deliver similar encapsulation efficiency (EE%). However, at 700 µL, TAMARA provides a clearly higher encapsulation yield (EY%), consistently exceeding 90%. By comparison, the toroidal mixer, even under optimized conditions at the same volume, achieves only around 75% yield. Reducing the working volume further decreases yield for both approaches, as noted above.
 Figure 7: Comparison of cell transfection efficiency, cell viability, and protein expression after 16 hours of exposure to TAMARA-formulated mRNA-LNPs at a 700 µL volume
Figure 7: Comparison of cell transfection efficiency, cell viability, and protein expression after 16 hours of exposure to TAMARA-formulated mRNA-LNPs at a 700 µL volume
Impact of formulation parameters on transcription efficiency, cell viability & protein expression
A: Impact of formulation parameters
Following physicochemical characterization of the RNA-LNPs produced with TAMARA, an in vitro study was completed using eGFP-encoding mRNA-LNPs to evaluate performance under biologically relevant conditions. This phase assessed how formulation parameters affect three key outcomes: translation efficiency (as an indicator of delivery performance), cell viability (to evaluate cytotoxicity), and protein expression (measured by mean fluorescence intensity per cell).
The data show that changes in formulation parameters had little to no impact on transfection efficiency, which remained close to 100%, or on cell viability, which stayed high with low cell death (<15%). However, protein expression levels did vary between samples. The markedly lower mean fluorescence intensity observed for the first sample (TAM 4-9 700) is most likely due to its lower encapsulation yield, as discussed previously. Differences among the remaining samples are likely driven by the combined effects of encapsulation yield, nanoparticle size, and other formulation parameters that influence cellular uptake.
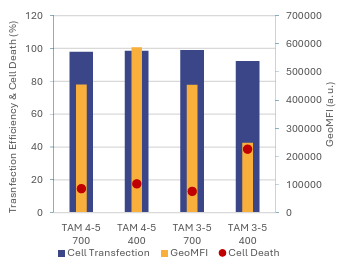
 Figure 8: Comparison of cell transfection, cell viability, and protein expression after 16 hours of exposure to mRNA-LNP formulated with TAMARA at 400 µL and 700 µL volumes (A), and at 400 µL and 200 µL volumes (B), under identical formulation conditions.
Figure 8: Comparison of cell transfection, cell viability, and protein expression after 16 hours of exposure to mRNA-LNP formulated with TAMARA at 400 µL and 700 µL volumes (A), and at 400 µL and 200 µL volumes (B), under identical formulation conditions.
B: Impact of formulation volume
Next, the researchers assessed how the total formulation volume affects performance, with the aim of defining the minimum practical working volume for in‑vitro studies. To do this, results obtained at different volumes were compared while keeping all flow conditions constant.
As shown in Figure 8, transfection efficiency remained high across all volumes, above 90% in every case. However, it declined slightly as the formulation volume decreased. Cytotoxicity was low at higher volumes but increased at smaller volumes.
These results indicate a clear relationship between encapsulation yield and cell expression: conditions that delivered higher encapsulation yields also produced higher cell expression levels. This is consistent with earlier findings showing that lower formulation volumes reduce encapsulation yield, which in turn lowers overall expression in cells.
C: Comparison between TAMARA & Toroidal mixer
Finally, RNA-LNPs formulated with TAMARA were compared to those produced using a toroidal microfluidic mixer operated under previously optimized conditions.
 Figure 9: Comparison of cell transfection, cell viability, and protein expression after 16 hours of exposure to mRNA–LNP
Figure 9: Comparison of cell transfection, cell viability, and protein expression after 16 hours of exposure to mRNA–LNP
formulations produced with TAMARA (all tested volumes and conditions) versus IGNITE (optimized conditions).
The data show that RNA‑LNPs produced with TAMARA and with the toroidal mixer deliver comparable transcription performance, with efficiencies close to 100%. Cell viability was similarly high in both cases, with only minimal cytotoxicity observed.
Notably, RNA‑LNPs formulated on TAMARA, even without prior optimization, achieved up to 30% higher protein expression than those produced on the optimized IGNITE system. This underlines the importance of both robust formulation platforms and systematic optimization to maximize protein expression. The results also confirm that formulation settings—specifically Total Flow Rate (TFR) and Flow Rate Ratio (FRR)—have a significant impact on in‑vitro performance, reinforcing the need for comprehensive screening of LNP formulation conditions.
DISCUSSION
What did the results show?
This study provides clear evidence that TAMARA is a highly effective and efficient system for formulating RNA-LNPs using microfluidics.
Both TAMARA and the toroidal mixer delivered strong physicochemical performance, meeting regulatory expectations (PDI < 0.2) and maintaining tight control over particle size. This confirms the suitability of microfluidic approaches for robust RNA-LNP production.
When different formulation volumes with TAMARA were examined, the 200 µL condition showed lower encapsulation yields and was more challenging to work with. In contrast, 400 µL and 700 µL runs delivered consistent, high-quality formulations. Based on this, 400 µL emerges as an ideal starting point for screening: it uses material efficiently while delivering reliable quality, and can then be scaled up for larger or in vivo studies.
In terms of encapsulation efficiency, TAMARA performed on par with the toroidal mixer. However, encapsulation yield clearly favored TAMARA. TAMARA delivered over 90% encapsulation yield at volumes as low as 400 µL. In comparison, the toroidal mixer achieved only around 75% at 700 µL, even under optimized conditions. This demonstrates TAMARA’s clear advantage for small-volume screening, enabling you to conserve costly RNA with minimal material loss.
In cell-based studies, both systems showed similarly high transcription efficiencies (close to 100%) and low cytotoxicity, indicating good safety and performance profiles. RNA-LNPs produced with TAMARA delivered up to 30% higher protein expression than those made with the previously optimized IGNITE system from Precision Nanosystems. This shows that TAMARA can match—and in some cases surpass—the performance of established toroidal microfluidic mixers for delivering functional RNA to cells.
Future work can focus on further reducing the minimum screening volume and using a structured Design of Experiments (DoE) approach to fine-tune TAMARA’s operating parameters. Even without extensive pre-optimization, TAMARA already outperforms toroidal mixers while significantly reducing RNA consumption. The entire TAMARA study was completed using only two chips, demonstrating excellent cost-efficiency for screening, and the system can be scaled to volumes in the tens of milliliters while maintaining the same high performance.
Conclusion
To summarize, the TAMARA system is a highly effective, and in many cases superior, alternative to Ignite by Precision Nanosystems and other toroidal micromixer technologies. It delivers consistently high performance across a broad range of volumes, while using less material and relying on reusable chips to reduce operating costs. This makes TAMARA a compelling, cost-efficient choice for advancing RNA‑LNP therapeutics research and improving overall accessibility.
SPECIFICATIONS
Material and methods
RNA-LNP Formulation
RNA-LNP composition
RNA: 5moU eGFP mRNA cleancap1
Lipid Composition: Proprietary LNP from ART “ARNm” lab
RNA-LNP formulation systems
In this study, the researchers used two separate nanoparticle formulation systems, each based on a different microfluidic mixing technology, to directly compare the performance of the two approaches.
TAMARA
TAMARA is a next-generation nanoparticle formulation platform developed by Inside Therapeutics.
This microfluidic platform supports the full R&D workflow for RNA-LNP medicines, from early screening through to in vivo studies. It uses a reusable chip with an optimized fluidic design that removes dead volumes, making Tamara a cost‑effective solution for RNA‑LNP development.
Within a single chip, TAMARA provides two selectable microfluidic mixing modes: an optimized Herringbone mixer and a Baffle mixer. For this study, the Herringbone mixer was used throughout.
Using the TAMARA system, RNA‑LNPs were formulated at different settings by adjusting the Total Flow Rate (TFR) and Flow Rate Ratio (FRR). Each batch was produced at a total volume of 200, 400, or 700 µL.
The table below summarizes the formulation conditions applied.
| Volumes (µL) | TFR (mL/min) | FRR |
| 700 | 9, 5 | 3:1, 4:1 |
| 400 | 5, 4 | 3:1, 4:1 |
| 200 | 4, 2 | 3:1, 4:1 |
The same formulation settings could not always be applied at every volume, as the formulation time became too short under low-volume, high–flow rate conditions. Importantly, the entire study was carried out using only two chips—one dedicated to the 700 µL batches, and one used for all 200 and 400 µL batches. Between each run, the chip was thoroughly cleaned using TAMARA’s built‑in cleaning cycle to maintain consistent performance and reliable, repeatable results.
IGNITE
Ignite, from Precision Nanosystems, is an industry‑leading system for nanoparticle formulation. It uses single‑use cartridges with integrated toroidal mixers to produce nanoparticles efficiently and consistently.
For this study, RNA‑LNP formulations on Ignite were pre‑optimized using a Design of Experiments (DoE) approach to define the best processing conditions. All formulations were prepared at a total volume of 700 µL.
Post formulation process
Before filtration, nanoparticles from each system were diluted in 9 mL of Milli-Q water and then processed using the standard filtration method.
Characterization
RNA-LNP size & PDI
RNA-LNP size and PDI were measured using a Zetasizer Ultra Red DLS system (Malvern Panalytical). Reported size values correspond to the z‑average of each sample. PDI (polydispersity index) ranges from 0 to 1 and indicates how uniform the LNP population is, with lower values reflecting higher homogeneity.
RNA-LNP encapsulation measurement
Encapsulation efficiency (EE%) and encapsulation yield (EY%) were measured using a modified RiboGreen assay and a Clariostar Plus plate reader.
EE% (encapsulation efficiency) is defined as the amount of RNA encapsulated divided by the total final amount of RNA.
EY% (encapsulation yield) is defined as the amount of RNA encapsulated divided by the initial amount of RNA used in the formulation.
Cell assays
T -1: Transfection Day -1:
Cells were seeded in the appropriate well plate, such that the cells were at 70-80% confluent on the end point.
T0: Transfection Day:
-
Media was removed from the cells
-
mRNA-LNP was added at a concentration of max 2 µg/mL of mRNA, 0.5 µg of mRNA per well.
-
Cells were incubated with the treatment for 24 hours
-
The mRNA–LNP formulation was prepared in Opti-MEM™ I Reduced Serum Medium, a transfection-optimized medium that allows reduced FBS usage. For overnight incubations, FBS was added to the Opti-MEM.
Flow cytometry
After transfection, protein expression was measured by flow cytometry (FACS) using the cells’ eGFP signal. The Geometric Mean Fluorescence Index (GeoMFI) was used to compare protein expression levels between conditions.
Before each FACS run, the culture media was replaced with FACS buffer. The FACS buffer contained: PBS without MgCl2, 5% FBS, 2.5 mM EDTA, DNase I (30 µg/mL), and 0.5% sodium azide.
GFP expression was quantified on a Cytoflex S cytometer. Cell viability was assessed by adding propidium iodide to the cells immediately before analysis.
References
1 Schober, G.B., Story, S. & Arya, D.P. A careful look at lipid nanoparticle characterization: analysis of benchmark formulations for encapsulation of RNA cargo size gradient. Sci Rep 14, 2403 (2024). https://doi.org/10.1038/s41598-024-52685-1
Get your free copy.
Open this application note in a downloadable format.
FURTHER RESOURCES
You may also be interested in these articles:
Brochure: TAMARA Nanoparticle Formulation System
Discover the key features and benefits of the TAMARA RNA-LNP formulation system. Link opens the brochure in a new window.
%20(1).webp?width=400&height=400&name=TAMARA-Flyer-Front-Cover-Widget%20(2)%20(1).webp) Explore
Explore
Blog: Introducing TAMARA: RNA-LNP formulation system
Microfluidics of IDEX MPT announces its partnership with Inside Therapeutics to distribute the TAMARA platform in the USA & Canada.
 Explore
Explore
Application Page: Lipid Nanoparticles
LNPs, including various lipid-based platforms such as liposomes, NLCs, and SLNs, are critical for the delivery of protein and amino acid-type antigens.
 Explore
Explore